 hydrogen gas (2 H-H) is combined with oxygen gas (O=O) to form two molecules of water in the reaction (2 H-O-H). \Ī chemical process is depicted in this diagram. Consequently, these gaps in the train lines compensate for the increase in the area. In the summer, the temperature rises, causing the metal tracks to expand. In light of this, it is clear why we see reduced spaces between rails on the railway lines. Metals expand when heated and compress when cooled, as we know. Following the heating procedure, the iron piece is pounded to the appropriate shape. The blacksmith, for example, can cast a piece of iron into a variety of shapes. Reversible and irreversible alterations both have their merits and disadvantages. Newer substances are formed when a substance undergoes a permanent change. The elongation of a spring or the folding of paper are two examples of reversible chemical changes.Īn irreversible change is a change that cannot be reversed by any physical or chemical methods. In the vast majority of circumstances, new material is not created when a chemical reaction is reversible. Only the physical qualities of the material, such as its size and shape, tend to change in a reversible alteration. One or more approaches can be used to undo this alteration. 
The term "reversible change" refers to a chemical alteration that can be undone. It is possible for a chemical alteration to be reversed, or it is not. Some instances of chemical alterations are the creation of dye in the colour and the chemical change in the colour of hair. 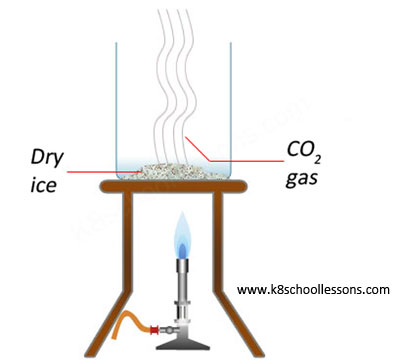
The oxidation reaction is one example of a chemical change that results in a chemical reaction.Ĭhemical reactions are normally irreversible, except in the case of reversible alterations in chemistry. Synthesis or breakdown of two substances might lead to a new substance being formed as a result of this process. One or newer substances are likely to form in this environment. This text is adapted from Openstax, Anatomy and Physiology 2e, Section 24.1: Overview of Metabolic Reactions and Openstax, Biology 2e, Section 2.1: Atoms, Isotopes, Ions and Molecules: The Building Blocks.One material can be transformed into another with differing qualities as a result of a chemical transformation. When there is excess hydrogen ions in the blood, forming carbonic acid will be the reaction's major direction. If we added carbonic acid to this system, some of it would convert to bicarbonate and hydrogen ions. However, biological reactions rarely obtain equilibrium because the concentrations of the reactants or products or both are constantly changing, often with one reaction's product a reactant for another. A chemical equation with a double-headed arrow pointing towards both the reactants and products often denotes these reversible reaction situations.įor example, in human blood, hydrogen ions (H +) bind to bicarbonate ions (HCO 3 -), forming an equilibrium state with carbonic acid (H 2CO 3). This back and forth continues until a certain relative balance between reactants and products occurs-a state called equilibrium. At this point, product and reactant designations reverse. In reversible reactions, reactants turn into products, but when the product's concentration goes beyond a certain threshold (characteristic of the particular reaction), some of these products convert back into reactants. In theory, any chemical reaction can proceed in either direction under the right conditions. Oxidation-reduction reactions often happen in a series so that a molecule that is reduced is subsequently oxidized, passing on not only the electron it just received but also the energy it received. These two reactions always happen together, and when an electron is passed between molecules, the donor is oxidized, and the recipient is reduced. The loss of an electron (oxidation) releases a small amount of energy both the electron and the energy are then passed to another molecule in the process of reduction or the gaining of an electron. A molecule gives up a hydrogen atom in the form of a hydrogen ion (H +) and an electron, breaking the molecule into smaller parts. The electrons in these reactions commonly come from hydrogen atoms, which consist of an electron and a proton.  
These reactions involve the transfer of electrons from one compound to another. An exchange reaction is a chemical reaction in which both synthesis and decomposition occur, chemical bonds are both formed and broken, and chemical energy is absorbed, stored, and released.Ī special kind of exchange reaction is the oxidation-reduction reaction, or the redox reaction. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
